Adjustment Abrasive Kit
Chairside Reline Cartridge System
Chairside Reline Powder & Liquid
Denture Accessories
Denture Adhesives
Denture Base Materials
High Spot Indicators
Miscellaneous
Orthodontic Resin
Repair Material
Separating Film
Temporary Crown and Bridge Material
Tissue Conditioner
Tray Materials
Calcium Hydroxide Cavity Liners
Cavity Varnished
Cement Accessories
Cement Cleaners
Desensitizers
Glass Ionomer Cavity Liners
Glass Ionomer Luting Cement
Permanent Zoe Cements
Polycarboxylate Cements
Resin Luting Cements
Surgical Cements
Temporary Crown & Bridge Cements
Temporary Filing Materials
Veneer Bonding System
Zinc Oxide Powder
Zinc Phosphate Cements
Absorbent Paper Point
Apex Locators
Calcium Hydroxide Points
Chelating Agents (Edta)
Endo Aspirator Tips
Endo Blocks & Ruler
Endo Inlays
Endo Medicaments
Endodontic Accessories
Endodontic Burs
Endodontic Handpieces
Endodontic Instrument
Endodontic Micromotors
Endodontic Organizers
Endodontic Sonic Instruments
Engine Files RA
Gutta Percha Points
Hand Files
Irrigating Needles & Tips
Irrigating Syringes
Irrigation Systems
NiTi Rotary Files
Obturation
Root Canal Sealers
Abrasive System
Air Compressor
Beneath Shelf Systems
Cart System
Chairs
Cuspidors
Delivery System
Delivery Systems
Economy Air Systems
Folding Arms/Brackets
Folding Vacuum Arm System
Handpiece Control Heads
Imaging Products & Accessories
Instrument Washer
Light Accessories
Mechanical Room Accessories
Mobile Cabinet Systems
Mounts/Bushings
Operatory Lights
Operatory Packages
Over The Patient Systems
Panel Mount Handpiece Controls
Portable Dental Unit
Post Mount Tray Tables
Stellar Cabinets
Stellar Delivery Console
Sterilizers
Stools
Telescoping/Folding Arms
Ultrasonic Cleaners
Vacuum Pumps
Water Distillers & Purifiers
Water System
X-Ray Processing Machine
Accessories & Parts
Air Free High Speed Handpieces
Automatic Cleaner & Lubricator System
Disposable Handpiece Systems
Electric Handpiece Systems
Fiber Optic Replacement Bulbs
Fiber Optics & Light Source System
High Speed / Fiber Optic
High Speed / Non Fiber Optic
High Speed / Surgical
Low Speed /Surgical
Low Speed /Surgical Electric Motors
Low Speed Handpieces and Parts
Lubricant
Replacement Turbines
Swivel Couplers
Accessories
Alcohol
Autoclave Tape and Label
Barrier Product
Biological Monitoring System
Cleaning & Hygiene for Hands
Disinfectants and Pre-Soaks
Disposable
Face Shields
Germicide Trays
Gloves
Masks
Nylon Tubing
Safety Glasses
Sterilization Pouches
Sterilization Wraps
Sterilizing Instruments
Surface Cleaners & Disinfectants
Alloy
Articulating Material
Blasters
Brushes
Buffs
Burs
Die Materials
Duplicating Products
Equipment
Felt Wheels, Muslin, Linen & Felts
Gypsums Products
HoneyComb Trays & Acessories
Investment
Lab Handpieces and Accessories
Lab Infection Control
Laboratory Accessories
Laboratory Instruments
Loupes & Visual Aids
MicroMotor
Modeling
Plastic Preform Patterns
Tin Foil Substitute
Torches and Burners
Waxes
Wire, Clasps and Acessories

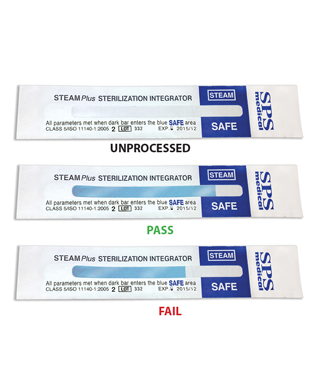
STEAMPlus Sterilization Class V Integrator, 11" x 8" x 7" - Pk/100
STEAMPlus Sterilization Integrator (SPS Medical)
STEAMPlus Type 5 Integrators
- For use in steam sterilizers
- Enhance sterility assurance protocol by monitoring every sterilization cycle
- Provides a definitive pass or fail result allowing for release of all non-implant loads before results of spore strip test are received
- FDA cleared as an equivalent in performance to a biological indicator, and offers the highest level of sterility assurance from a chemical indicator but as it does not contain live organisms to prove lethanlity does NOT replace the routine use/recommendations of biological monitoring
STEAMPlus Tray Record Card - When immediate use sterilization is unavoidable, utilize the Tray Record Card for documenting the sterilization cycle with patient traceability.
- The immediate readout STEAMPlus™ Sterilization Integrator is attached to a 3” x 5” card which allows for cycle parameters, load number and patient name to be recorded and kept as a permanent record.
- Release non implant loads immediately with peace of mind that parameters for sterilization have been met and maintain this card as a physical record of the sterilization cycle.
The STEAMPlus™ Sterilizer Test Pack has a STEAMPlus™ Sterilization Integrator inside a challenge pack for immediate load release of non implant gravity and prevacuum sterilization cycles.
- This product tests to ensure all parameters for sterilization have been met, and is FDA cleared as equivalent in performance to an AAMI BI Test Pack (PCD).
- For the highest level of sterility assurance, a STEAMPlus™ Sterilizer Test Pack should be run in every load.
- Loads containing implantable devices should include a BI Test Pack such as the SporViewPlus™
| MPN # | SSI-100 |
| Weight: | 3.00 lbs. |
| Price: | $45.50 |
Qty:
You may also interested in related products:
| MPN # | Product | Price, $ | |
|---|---|---|---|
| SSI-1000 |
STEAMPlus Sterilization Integrator Pk/1000 | 210.00 |
|
| TRC-050 |
STEAMPlus Tray Record Card Pk/50 | 25.80 |
|
| STP-025 |
STEAMPlus Sterilizer Test Pack Pk/25 | 126.30 |
|